
GUIDELINES
MANAGEMENT OF DENGUE INFECTION IN ADULTS
Mahiran Mustafa, Bee Kiau Ho, on behalf of the Development Group for Clinical Practice Guidelines on Management of Dengue Infection in Adults (Revised Second Edition)
A. Epidemiology
Dengue is one of the most important arthropod-borne viral diseases in terms of human morbidity and mortality. The number of reported dengue cases in Malaysia shows an increasing trend. Dengue Fever (DF) accounts for almost 95% of all reported cases. The percentage of serologically confirmed cases at the time of notification is relatively low due to lack of convalescent samples (second blood specimen) sent for confirmation. The incidence rate of dengue is highest among the working and school-going age groups. A total of 70-80% cases are from urban areas where there is a high density of population and rapid development activities which favour dengue transmission.
Dengue is caused by dengue virus and transmitted by Aedes aegypti and Aedes albopictus. There are four distinct serotypes, DEN-1, 2, 3 and 4. Each episode of infection induces a life-long protective immunity to the homologous serotype but confers only partial and transient protection against subsequent infection by the other three serotypes. Secondary infection is a major risk factor for severe dengue due to antibody-dependent enhancement. Other important contributing factors for it are viral virulence, host’s genetic background, T-cell activation, viral load and auto-antibodies.
B. Clinical Manifestations and Pathophysiology
Dengue is a systemic and dynamic disease. It is presented in three phases which are:-
|
|
|
The incubation period for the infection is 4-7 days (range 3-14 days). It may be asymptomatic or result in a spectrum of illness ranging from undifferentiated mild febrile illness to severe disease, with or without plasma leakage and organ impairment. In symptomatic cases, clinical, haematological and serological profiles changing from day to day. These changes accelerate by the hour or even minutes during the critical phase, particularly in those with plasma leakage.
i. Febrile phase
A case of dengue is suspected when a patient has an acute febrile illness with two or more of the following features:
|
|
|
|
|
|
|
|
|
OR |
|
|
|
||
The high grade fever usually lasts 2-7 days. Mild haemorrhagic manifestations like positive tourniquet test or petechiae and mucosal membrane bleeding may be seen in DF and Dengue Haemorrhagic Fever (DHF).
ii. Critical phase
Clinical deterioration often occurs in the critical phase and is marked by plasma leakage. This phase occurs after third day of fever or around defervescence (usually between third to fifth day of illness) when a rapid drop in temperature may coincide with an increase in capillary permeability. At this point, the patient will either become better if no or minimal plasma leak occurs, or worse if a critical volume of plasma is lost.
The critical phase lasts about 24-48 hours. Varying circulatory disturbances can develop. In less severe case, patient recovers spontaneously, or after a short period of fluid or electrolyte therapy. In more severe forms of plasma leakage, the patient may sweat, has cool extremities and prolonged capillary refill time. The pulse rate and diastolic blood pressure increase, and the pulse pressure narrows.
| Look out for clinical warning signs that may precede severe dengue or shock: | |
|
|
|
|
|
|
The patient can progress rapidly to profound shock and death if prompt fluid resuscitation is not instituted. The continuum of circulatory disturbances in dengue requires frequent monitoring of the patient.
Thrombocytopaenia and haemoconcentration (evidenced by a raised HCT from baseline or a drop in HCT after rehydration) are usually detectable before the subsidence of fever and the onset of shock. The HCT level correlates well with plasma volume loss and disease severity. However, it may be equivocal when there is frank haemorrhage, early and excessive fluid replacement or untimely HCT determination.
iii. Recovery phase
After 24-48 hours of defervescence, plasma leakage stops and followed by reabsorption of extravascular fluid. Patient’s general well being improves and haemodynamic status stabilises. During this phase, HCT level stabilises or drops further due to haemodilution following reabsorption of extravascular fluid.
C. Dengue Classification
The classification into levels of severity has a high potential for being of practical use in the clinicians’ decision as to where and how intensively the patient should be observed and treated (i.e. triage which is particularly useful in outbreaks). In 2009, World Health Organization (WHO) suggested a new Dengue Classification and Level of Severity as shown in Figure 1 below.
Figure 1: Suggested Dengue Classification and Level of Severity

Source: World Health Organization. Dengue Guidelines for Diagnosis, Treatment, Prevention and Control - New Edition 2009. WHO: Geneva; 2009
D. Laboratory Investigations and Diagnostic Tests
Diagnosis of dengue should be clinical with guidance from laboratory results. Leucopaenia followed by progressive thrombocytopaenia is suggestive of dengue infection. While a rising HCT accompanying progressive thrombocytopaenia is suggestive of DHF. There is no local data available on the normal range of HCT in adults. In the absence of a baseline HCT level, a HCT value of >40% in female adults and >46% in male adults should raise the suspicion of plasma leakage. Thus, serial full blood count (FBC) and HCT must be monitored (by using Dengue Monitoring Card) as the disease progresses.
Definitive diagnosis of dengue infection can only be confirmed in the laboratory. However, the interpretation of laboratory diagnostic results should be done in the clinical context. Laboratory confirmatory tests include antibody detection (serology), virus isolation, detection of virus genetic materials (polymerase chain reaction/PCR) and detection of dengue virus protein (NS1 antigen). It is recommended that:-
- If the dengue IgM is negative before day 7, a repeat sample must be taken in the recovery phase.
- Dengue non-structural NS1 antigen can be helpful in early phase (< day 5) of dengue infection.
E. Management
The management of dengue is symptomatic and supportive. The infection is a dynamic disease and management issues vary according to the three phases of the clinical course. It is crucial to recognise plasma leakage, shock and early or severe organ impairment. This can be achieved by frequent clinical and laboratory monitoring. The plan of management is:-
- All suspected dengue cases must be notified by telephone to the nearest health office within 24 hours of diagnosis, followed by written notification within one week using the standard notification form.
- If admission is indicated, before transfer:
- Stabilise the patient at primary care.
- Communicate with the receiving hospital/Emergency & Trauma Department.
- If admission is not indicated:
- Daily or more frequent follow-up is necessary especially from day 3 onwards until the patient becomes afebrile for at least 24-48 hours without antipyretics.
- Serial FBC/HCT must be monitored as disease progresses. Dengue patients who are managed in the outpatient setting should be provided with a disease monitoring record/card to ensure that all relevant information is available to all healthcare providers.
The decision for referral and admission must not be based on a single clinical parameter but should depend on the total assessment of the patient. Criteria for referral are:-
i. Symptoms |
|
|
|
|
|
|
|
|
|
|
|
ii. Signs |
|
|
|
|
|
|
|
|
|
|
|
iii. Special situation |
|
|
|
|
|||
|
|
||
|
|||
iv. Laboratory criteria |
|
|
|
|
|||
Patient who does not require admission must be advised to take adequate fluids (minimum of five glasses of isotonic drinks or juice). Intravenous (IV) fluid is indicated in patients who are vomiting, unable to tolerate oral fluids or having an increasing HCT (indicating on-going plasma leakage) despite increasing oral intake.
All patients with warning signs should be considered for monitoring in hospitals. Patients can be started with IV fluid 5-7 ml/kg/hour for 1-2 hours, then reduce to 3-5 ml/kg/hour for 2-4 hours, and then reduce to 2-3 ml/kg/hour or less according to the clinical response.
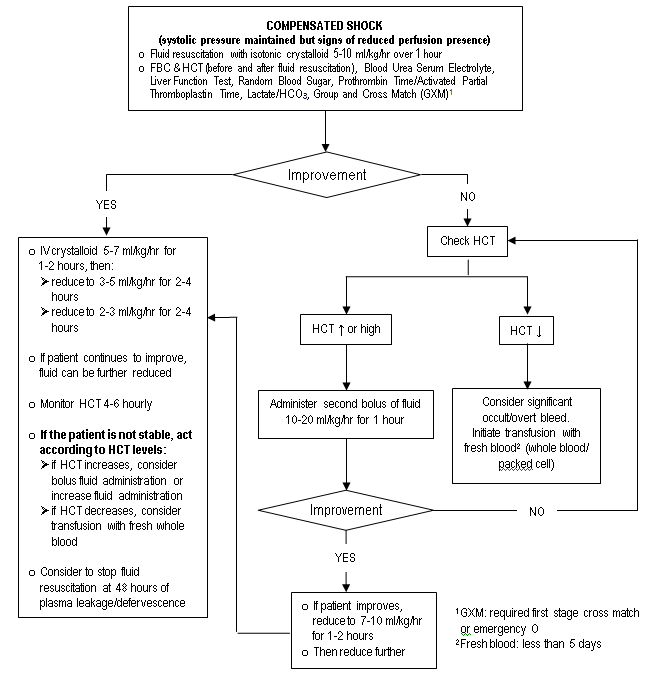
Crystalloid (such as 0.9% saline) is the fluid of choice for non-shock and Dengue Shock Syndrome patients (refer to Algorithm for Fluid Management in Compensated Shock). Reassess clinical status, repeat HCT and review fluid infusion rates accordingly. There is no role of prophylactic transfusion with platelets and fresh frozen plasma in dengue patients.
ALGORITHM FOR FLUID MANAGEMENT IN COMPENSATED SHOCK
ACKNOWLEDGEMENT
List of other members for the Development Group for Clinical Practice Guidelines on Management of Dengue Infection in Adults (Revised Second Edition): Abdul Hamid Jaafar, Ainul Nadziha Mohd Hanafiah, Chow Ting Soo, Faisal Salikin, Faraizah Abdul Karim, Mohd Ikhsan Selamat, Jameela Sathar, Lim Chew Har, Norita Ahmad, Salmiah Idis, Sheamini Sivasampu, Sin Lian Thye, K Sree Raman, Suresh Kumar, Tan Cheng Cheng and Tan Lian Huat.
Details of the evidence supporting these recommendations can be found in the CPG on Management of Dengue Infection in Adults (Revised Second Edition), available on the following websites: Ministry of Health Malaysia: http://www.moh.gov.my and Academy of Medicine: http://www.acadmed.org.my. Corresponding organisation: CPG Secretariat, Health Technology Assessment Section, Medical Development Division, Ministry of Health Malaysia & contactable at htamalaysia@moh.gov.my
